 
Even though the two dipole moments are equal in magnitude (since they are both oxygen hydrogen bonds), they are not arranged symetrically about the central atom and thus will not cancel out.\): Geometric structure of methyl isocyanate (CH 3–N=C=O), note there is no rotation around the double bonds only the single CN bond can rotate. Carbon dioxide will not be polar because both dipole moments are equal in magnitude (since they are both carbon oxygen bonds) and arranges symetrically about the central atom in a linear geometry. The purpose of this theory is to help visualize the chemical bonding of atoms in molecules. Water is a bent molecule with two polar bonds. The Lewis theory of chemical bondingalthough quite primitive and the most limited theory on electronic structuredoes help one to determine how valence electrons are arranged around the constituent atoms in a molecule. Carbon dioxide is a linear molecule with two polar bonds. Let's look at two examples: carbon dioxide and water. The general molecular geometry formula for N2 is A2X3N2. We know that N2 is the core atom, with six electrons pair bound (one NN) and two lone pairs of electrons. In the following geometries, the atoms are arranged symetrically about the central atom: linear, triangular planar, tetrahedral, square planar, triangular bipyrimidal, and octahedral. The lone pairs of electrons on the terminal nitrogen atom are denoted by the letter N. They are symetrically arranged about the central atom. BF3 Lewis Dot Structure VSEPR VB of high e- density regions around the central atom 3 atoms and 0 lone pairs the Electronic & Molecular Geometry. In the triatomic CO2 (carbon dioxide) molecule, the dipole moment is zero. The length of the carbon-oxygen bond in carbon dioxide is 116.3 pm, noticeably shorter than the roughly 140-pm length of a typical single CO bond, and shorter than most other CO multiply bonded functional groups such as carbonyls. Then draw the 3D molecular structure using VSEPR rules: Click and drag the. the bonds are made with the same element) ANDĢ. The symmetry of a carbon dioxide molecule is linear and centrosymmetric at its equilibrium geometry. Any resulting dipole indicates a polar molecule.ġ. Step 2: Using the molecular geometry, determine if any of the dipole moments will cancel. You have found all of the polar bonds in each of these three molecules. This is why H2O H 2 O doesnt have a perfect 109.5 109.5 angle between the bond pairs. Lewis structures for H 2S, BF 3, and CCl 2H 2Īre shown below. This occurs because of a difference in electronegativity of the two atoms that share the electrons. Predict the geometry of the following species: a) SO2 b). CO2 Describe the bonding in PCl5 without using d orbitals. Remember that a polar bond is one in which the electrons are unevenly distributed. Molecular Geometry: The molecular geometry of a compound is the molecular shape formed by all the atoms in the compound. Similarly two, C O bonds in CO2 molecule are counted as two bond pairs. Step 1: Indicate polar bonds in molecule or ion. Geometry of Molecules in which Central Atom has. As you work through these steps you will see that molecules with polar bonds are not necessarily polar molecules. In the CO2 Lewis structure, there are two oxygen atoms and one carbon atom.  
There is a series of steps you can take to determine if a molecule is polar or not. Carbon monoxide has a linear molecular geometry, a triple bond between C and O, and each atom contains one lone pair of electrons. 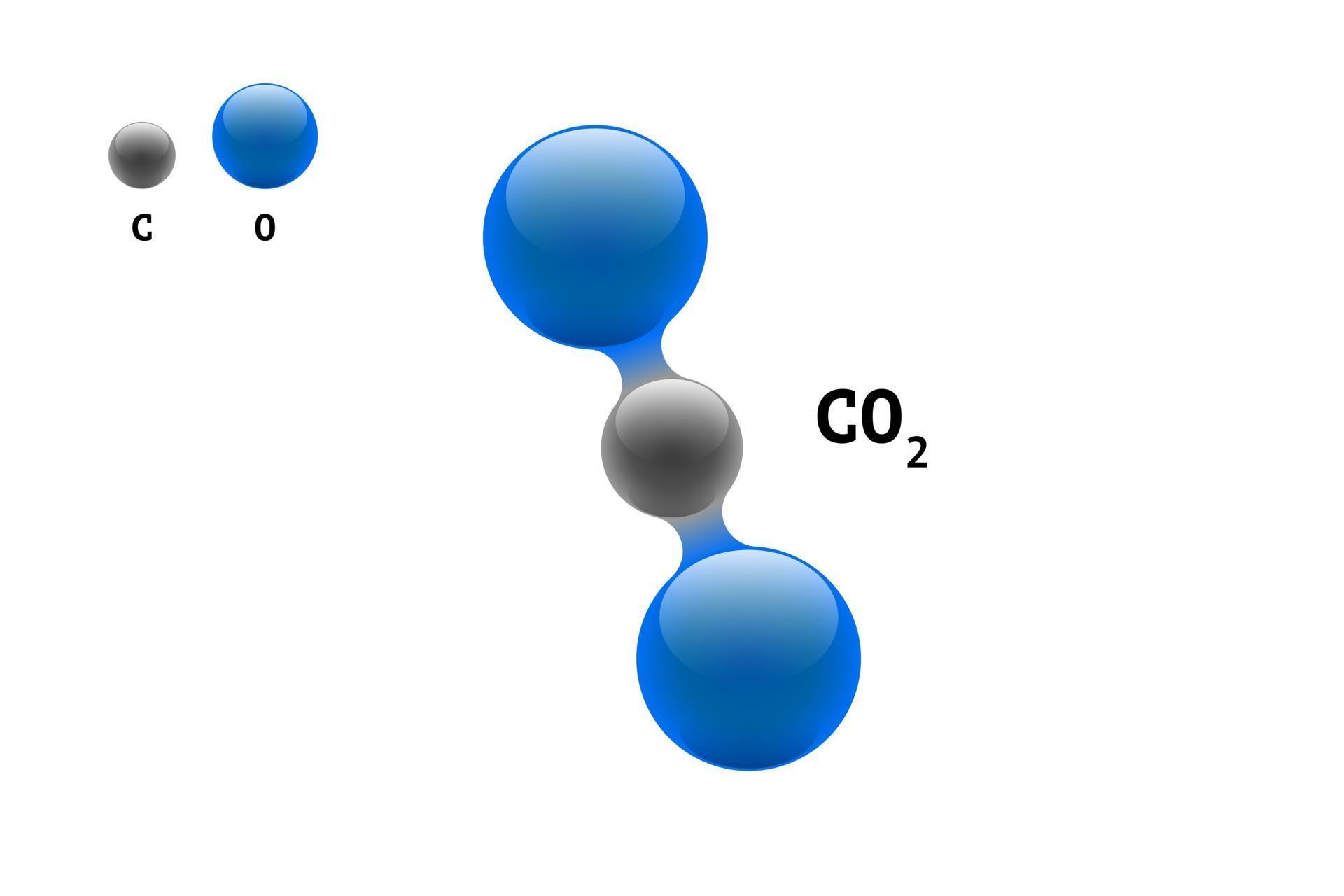
Likewise molecules in which there is an accumulation of electron density at one end of the molecule, giving that end a partial negative charge and the other a partial positive charge, are called polar molecules. The electron density of a polar bond accumulates towards one end of the bond, causing that end to carry a slight negative charge and the other end a slight positive charge. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
